Preparation of Silica Nano-Particles by Sol-Gel Method and Its Characterization
Neha Bhatt1, Abhilasha Mishra1,*, Rekha Goswami 2, 3 and Brijesh Prasad4
1Department of Chemistry, Graphic Era Deemed to be University, Dehradun, India
2Department of Environmental Science, Graphic Era Deemed to be University, Dehradun, India
3Department of Environmental Science, Graphic Hill University, Dehradun, India
4Department of Mechanical Engineering, Graphic Era Deemed to be University, Dehradun, India
E-mail: abhi1680@gmail.com
*Corresponding Author
Received 01 January 2021; Accepted 08 November 2021; Publication 30 November 2021
Abstract
Nano silica (SiO) has an extensive range of products from manufacturing to scientific. The Sol-gel technique in the field of material science is receiving worldwide interest because of its versatility for synthesizing silica nanoparticles. It provides a controlled composition of organic/inorganic hybrid material because of its unique and low-cost processing. The silica nanoparticles were prepared by the sol-gel method via two different routes first is a base-catalyzed hydrolysis reaction and the second is an acid-catalyzed hydrolysis reaction. The prepared silica nanoparticles were characterized by techniques Particle size analysis, Zeta potential, Fourier transform Infrared spectroscopy (FTIR), and X-Ray diffraction spectroscopy (XRD). Particle size analysis showed the nano range of particle and zeta potential proves the stability of prepared sols. Observed peaks from IR spectra are confirming the presence of a silica network. The amorphous silica was verified by the absence of sharp peaks and the presence of a broad peak at a 2 angle of 23.8 for the XRD analysis structure in the produced NPs.
Keywords: Silica nanoparticle, Sol-gel, acid hydrolysis, base hydrolysis.
1 Introduction
Nanotechnology is known for methods and techniques to prepare and improve material with novel functionalities and characteristics. Nanoparticles (NPs) are useful in different industries due to their unique and interesting properties different from their bulk material. Silica NPs are most attracted, among various types of NPs because of their simple functionality and controllable particle size and shape according to requirement. It is the class of nanoparticles that is most studied and investigated. Nano-sized silica nanoparticle shows some unique properties like low toxicity, high chemical inertness, high optical transparency, and excellent biocompatibility, which makes them eligible for a wide range of applications in industrial, medical, and chemical purposes. SiO nanoparticles have applications in electronics, thin films, electrical, insulators, and sensors, etc. (Park et al., 2002). Synthesized silica nanoparticles by sol-gel methodology are famous for their features like high purity, porosity, homogeneity, thermal stability, high rigidity, nano-scale structuring, high photochemical and thermal stability, and great optical transparency, which make it sensitive, selective, and suitable to incorporate in several applications (Wen et al., 1996). Colloidal silica is a source of fine silica nanoparticles with high surface area with innumerable applications such as binders, refractors, anti-soiling, reinforcing agents for adhesives, anti-blocking agent for polymers, etc (Azlina et al., 2016; Singh et al., 1999). Nano-dispersed silica particles are also used in various fields of chemistry like chromatography, ceramics, pigments, catalysts, etc because of their various properties like a uniform in size and shape (Do Kim et al., 2002). Silica nanoparticles are applicable in hydrophobic coating other than other nanoparticles due to their thermal and mechanical stability that can make long-lasting stable coating with a high amount of surface roughness. The superhydrophobicity can be achieved by on a rough nanoparticle-coated surface, layering of the low energy surface. Due to environmental stability and durability silica porous coatings are also used for antireflective coatings for glass (Kesmez et al., 2011).
Silica Nanoparticles are porous in structure so that they can regulate drug release according to different ranges of pH, temperature, light, and biomolecules. Meso-porous silica nanoparticles have been successfully used as gene transfection carriers, controlled drug delivery systems, in vitro targeting, therapy, imaging, and to deliver hydrophobic drugs. These are also used as an encapsulating agent with combinations of the other organic doping dyes and used as barcoding tags. Silica nanoparticles are also useful in cancer therapeutics (Cholkar et al., 2017; Thakur et al., 2021). Due to properties, like biocompatibility and biodegradability, silica nanoparticles have been used as fillers, binders in medicine, coatings, rubber, plastics, as an additive in food, pharmaceutical, and cosmetic products, and as starting materials for semiconductors, silicates, and ceramics. (Mebert et al., 2017; Stephen et al., 2021). Their biodegradability, high mechanical strength enhances the ability to stimulate tissue repair. Silica nanoparticles are known to be safe for the conjugation of stocktickerDNA and drug delivery. Silica nanoparticles show greater absorption capacity by the plant cells that’s why used for increasing the seed viability of many plants. Meso-porous silica, also used for various catalytic processes. Silica nanoparticles increase nutritional content in pre-treated seeds due to the increased absorption capacity and show a better germination rate. Silica nanoparticles do not show any adverse effect on the plant even they improve the strength of the plants and structural rigidity of the plants (Jeelani et al., 2020).
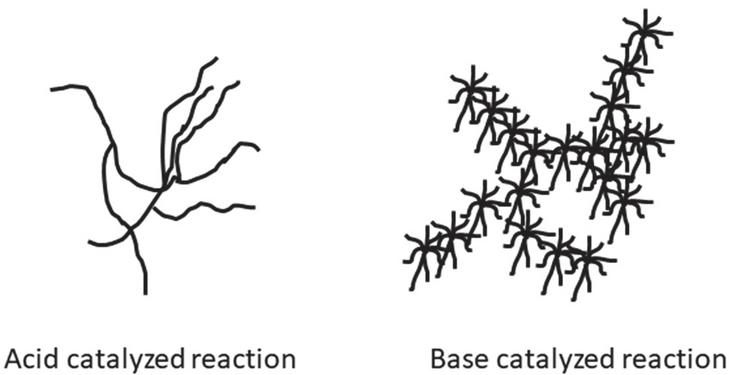
The sol-gel method is the best method for the preparation of nanoparticles and this method is extensively used for the preparation of nanoparticles because of very ease of processing, low-temperature process, and controlled morphology of particles. In acid-catalyzed reaction, the growth of the poly-silicates occurs in weakly branched by aggregation of clusters. In base-catalyzed sols, the poly-silicates formed are highly branched and compact polymer clusters. Acid-catalyzed sols degree of deprotonation is lower as compared to base-catalyzed reaction. This is the cause of high branching in the base-catalyzed system. The branching type in acid and base-catalyzed sols is shown in Figure 1 (Mehner et al., 2010).
Figure 1 Silica nanoparticle shape in acid-catalyzed and base-catalyzed reaction.
Uniform and transparent, single or poly-component ceramics and glasses, can be obtained at low temperatures from the metal-alkoxides solutions (Saka et al., 1987). Silica nanoparticles are also used for silica sol-gel coatings- It is used as a protective coating on metal sheets, Micro-patterning on glass substrates, used for corrosion protection, drag reduction, antireflective coating, dust repellent like purposes (Minami et al., 2013; Wen J et al., 1996). Silica nanoparticles in form of coatings and encapsulated silica nanoparticles with polymer are used for various applications such as drug delivery, etc (Chen et al., 2014). Deepa sriramulu et al. prepared silica nanoparticles by using a reverse microemulsion method for Superhydrophobic coating and monodispersed silica nanoparticles of 40 nm were obtained (Sriramulu et al., 2016). Spherical silica nanoparticles of size 80 nm–150 nm were prepared by the sol-gel method by LP Singh et al. and used to increase cement relating reactions (Singh et al., 2011).
In this paper, we report a sol-gel method for the preparation of metal-oxide nano-particles from their precursor by the acid and base-catalyzed process.
2 Materials and Method
Tetraethoxyorthosilicate (TEOS), Ammonium hydroxide (NHOH), Ethyl alcohol (EtOH), Hydrochloric acid (HCl). All Chemicals used were of Laboratory-grade reagent.
3 Experimental Section
3.1 Preparation of Silica Sol and Nano-powder by Base Catalysis
Silica precursors tetraethoxysilane (TEOS) was taken and mixed with ethyl alcohol (EtOH) in the ratio of 1:1. After that 1 N ammonium hydroxide (NHOH) was added slowly and drop-wise manner to initiate and catalyze the reaction. For hydrolysis, the solution was stirred for two hours at a constant speed of 400 rpm at room temperature. This solution was kept at room temperature for four days for the completion of the reaction. After completing the reaction, the nano-dispersed silica sol was prepared. This sol solution can be directly applied to the application area or further converted to gel and dry nano-powder. Then the prepared sol solution was heated up to 250C for 3–4 hours to complete condensation the sol solution was converted to silica nano-powder.
3.2 Preparation of Silica Sol and Nano-powder by Acid Catalysis
Silica precursors tetraethoxysilane (TEOS) was taken and mixed with ethyl alcohol solvent in the ratio of 1:1 and metricconverterProductID1 M1 M, hydrochloric acid (HCl) was added drop-wise to initiate and catalyze the reaction. The stirring of the solution was done for two hours at a constant speed of 400 rpm at room temperature for complete hydrolysis. This reactant mixture was kept in the fridge to prevent gel formation because hydrolysis reaction is more rapid in acid catalysis compared to base-catalyzed reaction. After completion of the reaction, the nano-dispersed silica sol was ready. This acid-catalyzed sol is easily converted to gel in a few days. The prepared gel was heated to 250C for 3–4 hours to make silica nano-powder.
The experimental process of preparation of nanoparticles in the form of sol and dry nanopowder is shown in Figure 2.
Figure 2 Preparation steps of sol and gel of nano-silica by sol-gel methodology.
3.3 Mechanism of Base-catalyzed Hydrolysis of Silica Sol During Preparation
In the base-catalyzed reaction hydroxyl ions (OH-) of the base being strong nucleophile attack directly on electropositive silicon atom of precursor and replaces –OR group. This associative mechanism forms a pentacoordinated intermediate. This is an SN type of reaction.
3.4 Mechanism of Acid-catalyzed Hydrolysis of Silica Sol During Preparation
The acid-catalyzed reaction is electrophilic. In acid catalysis, the protons present in the solution attack at the oxygen atom of the Si– OR group. After the attack, the positive charge on the silicon atom was increased. Now silicon becomes more reactive because of its more electrophilic nature.
4 Characterization
Prepared nano-silica sol and nano-powder from two different catalytic procedures acid-catalyzed reaction and the base-catalyzed reaction was characterized by Fourier Transform Infrared (FTIR) spectroscopy and X-Ray diffraction (XRD) spectroscopy.
4.1 Particle Size Analysis and Zeta Potential
The particle sizes were analysis and zeta potential analysis of prepared acid and base-catalyzed nano-silica sols were done by Instrument – Microtrac, Model – Nanotrac, wave II.
For determining the particle size of nanoparticles particle size analysis was done. To determine stability, and distribution of surface charge of nanoparticles in nano-colloidal solution, the zeta potential of nanoparticles was observed.
Zeta potential is known as the potential between the charge of nanoparticles and the formed layer of charge which was formed over the surface. The magnitude of zeta potential signifies the stability of the colloidal solution.
4.2 Fourier Transform Infrared Spectroscopy (FT-IR)
Both Prepared sols were characterized by FTIR spectroscopy. The principle of IR spectroscopy is when the IR rays pass through the sample, absorption, reflection, or transmittance is observed and an IR spectrum is formed. The resulting IR spectrum of the compound is unique for a particular sample and known as molecular fingerprint spectra of the compound. It helps to determine functional groups, bonding structure, and organic groups. The FTIR spectra of prepared sols were analyzed by Perkin Almer Spectrum two.
4.3 X-Ray Diffraction Spectroscopy
Both base-catalyzed and acid-catalyzed prepared nanoparticles were characterized by X-ray diffraction spectroscopy. X-ray diffraction (XRD) measurement was carried out by the instrument named Rigaku MiniFlex 600 x-ray diffractometer at IIT Kanpur.
5 Result and Discussion
Silica sols were prepared by the sol-gel method via the routes of base-catalyzed hydrolysis and acid-catalyzed hydrolysis. The prepared nanoparticles were characterized for confirmation test by FTIR and XRD respectively.
5.1 Particle Size Analysis and Zeta Potential
Both kinds of silica sols showing a nano-range of particles having an average size of the particle is 61 nm and 85 nm for base-catalyzed and acid-catalyzed silica sols respectively. Particle size analysis confirms the nano dimensional particles are present in the sol. Zeta potential is 137 mv and 104 mv for the acid-catalyzed and base-catalyzed sols, which indicates the very high stability of prepared silica nano sol.
5.2 Fourier Transform Infrared Spectroscopy (FTIR)
The IR analyzed spectra/curves of both kinds of sols are shown in Figures 3 and 4 respectively.
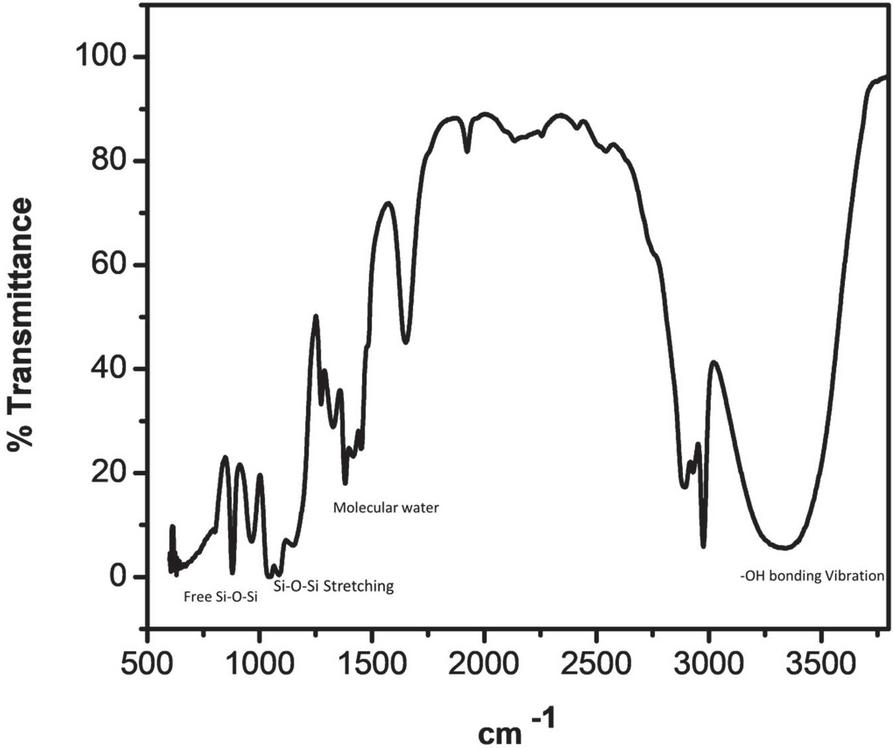
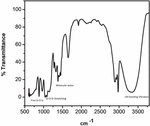
Figure 3 FTIR spectra of base-catalyzed silica sol.
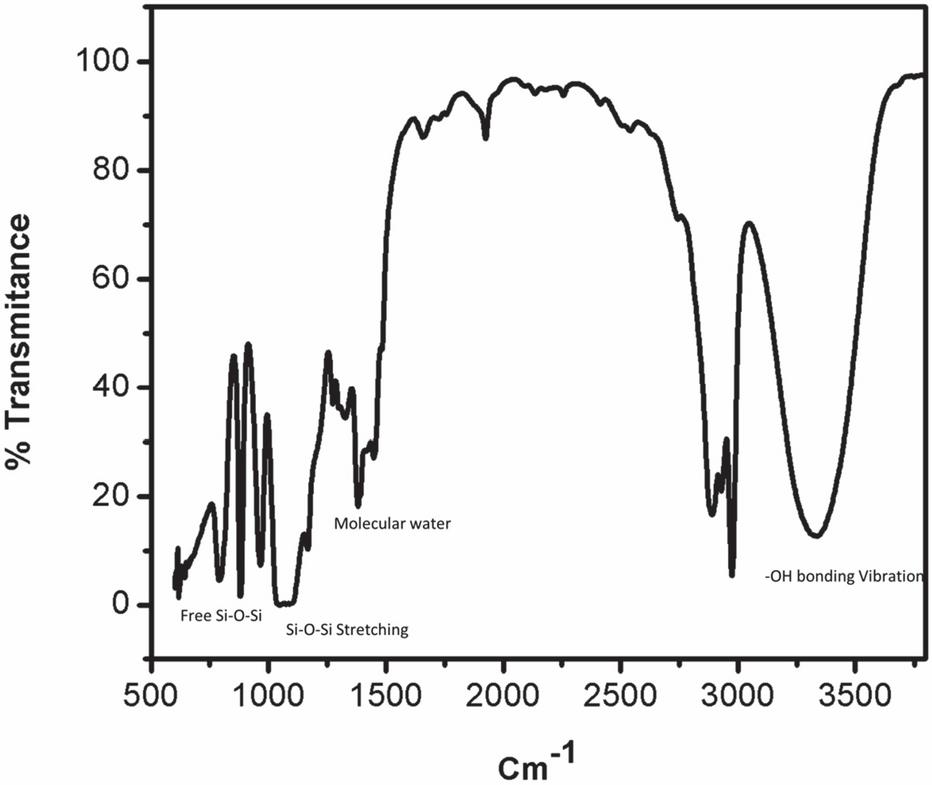
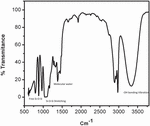
Figure 4 FTIR spectra of acid-catalyzed silica sol.
At around 3400 cm and 1630 cm broad peaks were appeared, because of molecular water and –OH bonding vibrations. At 3400 cm broader peak appeared in the base-catalyzed reaction, this is because of a more concentrated structure present, which makes large hydrogen bonding as compared to acid-catalyzed reaction, where less H bonding because of less branched structure.
Peaks at near 1100 cm and 480 cm are associated with stretching and out-of-plane vibrations of Si-O-Si bonds. The IR absorption band at 800 is due to vibrations of SiO. The band appeared near 470 cm is due to the out-of-plane deformation of Si-O bonds.
The band around 950 cm represents the stretching vibrations of the free silanol (Si-O-Si) group of silica networks present in the sol solution. The bending vibration of the C-H of the CH-O-Si group occurs at 790 cm. Major peaks are shown in Figures 3 and 4.
5.3 X-Ray Diffraction Spectroscopy
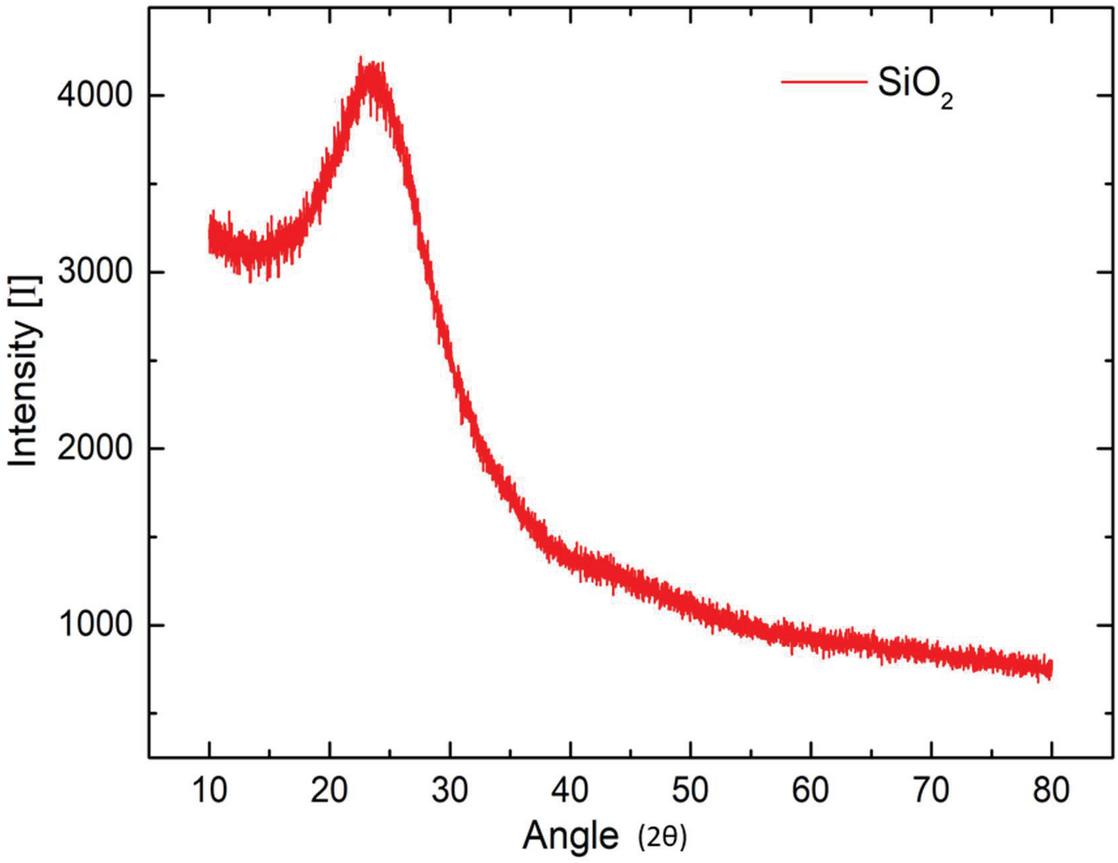
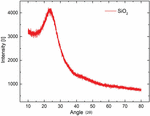
Radiation of wave in the 2 range from 10 to 80 for both samples. The observed X-RAY diffraction spectra between the intensity of radiation and 2 angle is shown in Figure 5 for base-catalyzed nanoparticles and in Figure 6 for acid-catalyzed silica nanoparticles. A broad peak at a 2 angle of 23.8 is recorded for both acid-catalyzed and base-catalyzed silica nanoparticles. Observed XRD pattern of prepared silica nanoparticles reveals that silica prepared by both methods is amorphous in nature and represents the silica (SiO). The acid-catalyzed reaction shows broader peaks, which means more amorphous behavior is observed compared to base hydrolyzed silica nanoparticles.
Figure 5 XRD pattern for base-catalyzed silica nanoparticle.
Figure 6 XRD pattern for acid-catalyzed silica sol.
6 Conclusion
Silica (SiO) nanoparticles were prepared successfully by acid-catalyzed reaction and the base-catalyzed reaction of sol-gel methodology. Nano-silica sols have a nano dimensional particle range showing high stability in solution. The confirmation of particles was also done by FTIR and XRD analysis. Prepared silica nanoparticles in the form of nano-sols and nano-powder can be further used for various applications.
References
Azlina, H.N., Hasnidawani, J.N., Norita, H. and Surip, S.N. (2016). Synthesis of SiO nanostructures using sol-gel method. Acta Phys. Pol. A, 129(4), 842–844.
Chen, D., Singh, D., Sirkar, K.K., Zhu, J. and Pfeffer, R. (2014). Continuous polymer nanocoating on silica nanoparticles. Langmuir, 30(26), 7804–7810.
Cholkar, K., Hirani, N. D., and Natarajan, C. (2017). Nanotechnology-based medical and biomedical imaging for diagnostics. In Emerging nanotechnologies for diagnostics, drug delivery and medical devices (pp. 355–374). Elsevier.
Do Kim, K. and Kim, H.T. (2002). Formation of silica nanoparticles by hydrolysis of TEOS using a mixed semi-batch/batch method. Journal of sol-gel science and technology, 25(3), 183–189.
Jeelani, P.G., Mulay, P., Venkat, R. and Ramalingam, C., (2020). Multifaceted application of silica nanoparticles. A review. Silicon, 12(6), pp. 1337–1354.
Kesmez, Ö., Burunkaya, E., Kiraz, N., Çamurlu, H.E., Asiltürk, M. and Arpaç, E. (2011). Effect of acid, water and alcohol ratios on sol-gel preparation of antireflective amorphous SiO coatings. Journal of Non-Crystalline Solids, 357(16–17), 3130–3135.
Mebert, A.M., Baglole, C.J., Desimone, M.F. and Maysinger, D. (2017). Nanoengineered silica: Properties, applications and toxicity. Food and Chemical Toxicology, 109, pp. 753–770.
Mehner, A., Dong, J., Prenzel, T., Datchary, W. and Lucca, D.A. (2010). Mechanical and chemical properties of thick hybrid sol–gel silica coatings from acid and base catalyzed sols. Journal of sol-gel science and technology, 54(3), 355–362.
Minami, T. (2013). Advanced sol–gel coatings for practical applications. Journal of sol-gel science and technology, 65(1), 4–11.
Park, S.K., Do Kim, K. and Kim, H.T. (2002). Preparation of silica nanoparticles: determination of the optimal synthesis conditions for small and uniform particles. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 197(1–3), 7–17.
Sakka, S. (1987). Sol-gel glasses and their future applications. Transactions of the Indian ceramic society, 46(1), 1–11.
Singh, L.P., Agarwal, S.K., Bhattacharyya, S.K., Sharma, U. and Ahalawat, S. (2011). Preparation of silica nanoparticles and its beneficial role in cementitious materials. Nanomaterials and Nanotechnology, 1, 9.
Singh, T.K., Jain, C.L., Sharma, S.K. and Singh, S.S.(1999). Preparation of dispersed silica by hydrolysis of tetraethyl orthosilicate.
Sriramulu, D., Reed, E.L., Annamalai, M., Venkatesan, T.V. and Valiyaveettil, S. (2016). Synthesis and characterization of superhydrophobic, self-cleaning NIR-reflective silica nanoparticles. Scientific reports, 6(1), 1–10.
Stephen, S., Gorain, B., Choudhury, H. and Chatterjee, B. (2021). Exploring the role of mesoporous silica nanoparticle in the development of novel drug delivery systems. Drug delivery and translational research, pp. 1–19.
Thakur, C., Nayak, P., Mishra, V., Sharma, M. and Saraogi, G.K. (2021). Treating blood cancer with nanotechnology: A paradigm shift. In Nano Drug Delivery Strategies for the Treatment of Cancers (pp. 225–243). Academic Press.
Biographies

Neha Bhatt is research scholar at Graphic Era Deemed to be University since 2018. She has done her master in Chemistry from University of Delhi in 2015. Her research areas are nanomaterials, synthesis of different material and nanostructured coating, super-hydrophobic antireflective coating.

Abhilasha Mishra presently working as Associate Professor in Chemistry Department of Graphic Era Deemed University, Dehradun. She has compleated her MSc. From Chemistry department RDVV University in 2002 and done her Ph.D. from RDVV university in 2007. She is having 14 years of Teaching and research experience. Her research areas includes nanomaterials, nanostructured smart coatings, biomaterials, water remediation using nanomaterials and solar cell efficiencies.

Rekha Goswami is research scholar at Graphic Era Deemed to be University since 2019. She has done her master in Environmental Science from HNB Garhwal University in 2014. Her research areas are nanomaterials, nanocellulose membrane and waste water treatment.

Brijesh Prasad is associated with the Department of Mechanical Engineering at Graphic Era Deemed to be University, Dehradun Uttarakhand, India as an Assistant Professor. Dr. Brijesh Prasad, presently working as a postdoc fellow with Institute of Advanced Materials, IAAM (Sweden). His research area covers the development of smart nanocomposite materials for energy harvesting and sensing applications for soft electronic devices.
Journal of Graphic Era University, Vol. 9_2, 215–230.
doi: 10.13052/jgeu0975-1416.927
© 2021 River Publishers